Pilihan
AWG Kibarkan Bendera Indonesia-Palestina di Gunung Raung
Pulanglah, Ali…
Alamar Biosciences Launches NULISAseq Neuro 220 Panel, its largest precision proteomics panel with the sensitivity and specificity to advance neurological disease research
New panel expands coverage for Alzheimer’s and Parkinson’s diseases including 15 new biomarkers developed with support from The Michael J. Fox Foundation for Parkinson’s Research (MJFF) and 12 assays specific to brain and peripheral Tau isoforms
NULISA data to be presented in over 11 scientific sessions and 25 posters at the International Conference on Alzheimer’s & Parkinson’s Diseases (AD/PD 2026)
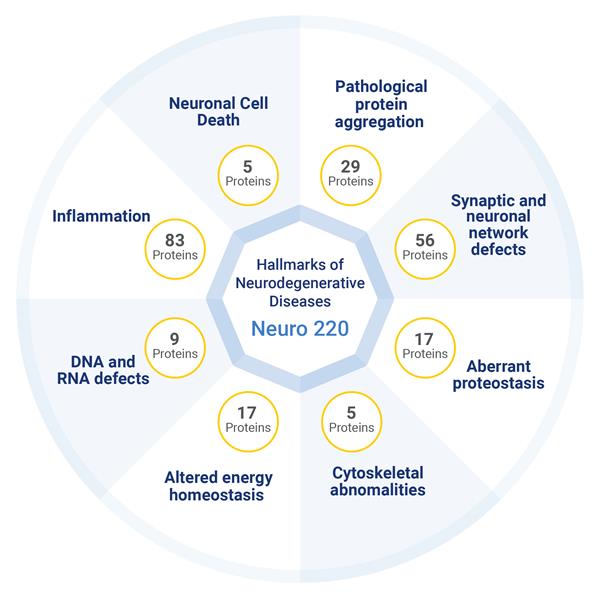
FREMONT, Calif., March 17, 2026 (GLOBE NEWSWIRE) -- Alamar Biosciences, Inc. (“Alamar”), a leader in precision proteomics dedicated to advancing the early detection of disease, is proud to announce the launch of the NULISAseq Neuro 220 Panel with multiplexed measurement of 220 biomarkers from a single sample while maintaining ultra-high sensitivity and high specificity.
The panel expands on the flagship NULISAseq CNS Disease Panel 120 and features new biomarker content developed with support from MJFF for Parkinson’s Research as well as a substantial collection of highly selective Tau protein assays targeting both brain-derived and peripheral isoforms of total tau and the phosphorylated species; pTau217, pTau181, pTau205, pTau212, and pTau231. We believe this represents the largest suite of Tau assays available in a single panel and demonstrates the ability of the NULISA chemistry to distinguish between organ specific isoforms as well as a variety of post translational modifications.
“We are proud to support Alamar as they work to develop highly sensitive assays for several known and emerging biomarkers important in Parkinson’s disease research,” said Nicole Polinski, PhD, Director, Research Programs at MJFF. “We believe these assays will help the research community to better understand the progression of disease and expedite the development of diagnostic and prognostic signatures.”
“This launch reflects our continued innovation in precision proteomics, expanding our neurodegenerative disease panel to better capture the molecular complexity of diseases such as Alzheimer’s and Parkinson’s,” said Dr. Yuling Luo, founder, chairman, and chief executive officer of Alamar. “We’re grateful for The Michael J. Fox Foundation’s trust and partnership, and for their collaboration in advancing Parkinson’s novel biomarker content for the research community.”
The NULISAseq Neuro 220 Panel raises the standard for neurodegenerative disease research, applicable to a broad range of neurological disorders including Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, frontotemporal dementia, Lewy body dementia, progressive supranuclear palsy, amyotrophic lateral sclerosis, Huntington’s disease, vascular dementia, gliomas and traumatic brain injury. The panel’s ultra-high sensitivity and high specificity ensure compatibility with non-invasive sample collection devices and supports pre-symptomatic detection, while automated workflows using the ARGO HT System deliver exceptional reproducibility and ease of use, facilitating the analysis of disease heterogeneity, detection of co-pathologies, and the identification of prognostic or therapeutic response signatures across clinical cohorts.
The panel will be featured at AD/PD 2026, taking place from March 17-21, in Copenhagen, Denmark. Dr. Henrik Zetterberg will present data demonstrating the utility of the NULISAseq Neuro 220 Panel in neurodegenerative diseases during Alamar’s product theater on March 18.
"The ability to measure a wide array of neurodegenerative biomarkers in a single, highly sensitive assay represents a significant leap forward for both translational and clinical research," said Henrik Zetterberg, MD, PhD, Professor of Neurochemistry at University of Gothenburg. "Innovations like the NULISAseq Neuro 220 Panel empower us to better understand disease mechanisms and accelerate the development of effective diagnostics and therapies."
Additional data illustrating the utility of the NULISA platform for ultra-high sensitivity multiplexed detection of critical neurological biomarkers will be presented in over 11 oral presentations and 25 posters at AD/PD 2026.
For more information about the NULISAseq Neuro 220 Panel and Alamar’s full portfolio of precision proteomic solutions, visit alamarbio.com.
About Alamar Biosciences, Inc.
Alamar Biosciences is a privately held life sciences company dedicated to powering precision proteomics to enable the earliest detection of disease. Leveraging its proprietary NULISA technology and the ARGO HT System, Alamar’s platform is designed to deliver ultra-high sensitivity and address key limitations of existing technologies, helping researchers unlock the full spectrum of protein biomarkers across disease states. For more information, please visit alamarbio.com.
Forward Looking Statements
This press release may contain forward-looking statements, including statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. These include, without limitation, statements regarding Alamar Biosciences’ participation at upcoming conferences. Alamar Biosciences explicitly disclaims any obligation to update any forward-looking statements except to the extent required by law.
Media Contact:
media@alamarbio.com
Investor Contact:
investors@alamarbio.com
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/8ce2de83-a4b0-4378-b01f-03223b516e97
Copyright © 2026 GlobeNewswire, Inc.

.png)

Berita Lainnya
Q2 Metals Announces Inferred Mineral Resource Estimate on the Cisco Lithium Project with 295 Million Tonnes Grading 1.36% Li2O
HYCU Expands R-Shield Cyber Resilience Solution with Halcyon to Deliver the First Ransomware-Resilient Data Protection Solution
Hainan Tourism Promotion Seminar Lands in Madrid, Paving the Way for Deeper Cultural and Tourism Ties Between China and Spain
HKC and ANTGAMER Wrap Up 2026 HKTDC Fair as Fate Trigger Collaboration Draws Strong Attention
Crossing the Privacy Divide: Dmitry Shubov Speaks on How Southeast Asian Legal-Tech Founders Can Confidently Enter the U.S. Market
Philips receives FDA 510(k) clearance for Verida, the world's first AI-powered detector-based spectral CT*, advancing diagnostic precision across clinical applications
Boehringer Ingelheim delivers on late-stage pipeline with two key launches, grows sales by 7.3%* in a successful 2025
HERE Technologies dan MengQing memperluas penerapan navigasi cerdas untuk produsen kendaraan komersial terkemuka di Tiongkok.
Omni Bridgeway Appoints Peter Galgay as Head of Commercial Strategy and Capital Solutions
HKC and ANTGAMER Wrap Up 2026 HKTDC Fair as Fate Trigger Collaboration Draws Strong Attention
Zymeworks to Present Clinical and Preclinical Data on ADC Programs Including Novel RAS ADC Platform at AACR Annual Meeting
NorthStar Earth & Space Announces Plans to Become Publicly Traded via Merger with Viking Acquisition Corp. I